 If there is a method to physically separate the components (like boiling, evaporation, distillation, magnetism, or filtration), then a physical change has occurred. If composition is not changed, then this is a physical change. If you add HCI and it didnt precipitate, and in flow chart 2 you test with Blue litmus and it turned red, and when you add sodium sulfide and it has a yellow. We also provide students in other disciplines with an introduction to the principles and practices of this central. Our broad range of courses offers a rigorous core education in the theory and practice of chemistry. The physical changes are salt dissolving water, copper conducting, and water boiling. The Department of Chemistry at Saint Mary’s University offers undergraduate education in a supportive and friendly environment. Answer bĪ heterogeneous mixture is obviously a mixture, such as dirt a homogeneous mixture behaves like a single substance, such as saltwater. Our toilets do use greywater which can look heterogeneous (ok for sewer but not to drink). Tata Chemical Futures Intraday/Short Term Post a fantastic move stock is consolidating in a range. Natural and man-made chemicals are in our tap water (trust me, you want the chlorine). Lastly, Furman tap water is usually a homogeneous mixture. FLOWCHART 1: Identification of Unknown Nonmetal Anion Using Chemical Precipitation Tests - Refer to your Data Table to complete the chart. Furman Lake is definitely a heterogeneous mixture (in some areas it can get a bit swampy). A matter flowchart is a diagram usually used widely in chemistry to classify matter. Overview Definition And Purpose of Matter Flowcharts Definition. Qualitative data analysis often utilizes flow charts to demonstrate specific movement within a complex system. If there is no burning or dust build-up in the area, this is safe to assume. With the help from the flow chart, everyone can easily take away some basic ideas about this subject, and the method to create a matter flowchart from scratch will be discussed below. Hopefully, Furman air is a homogenous mixture (solution). Which is/are physical change(s): burning of fuel, digestion of food, salt dissolving in water, copper conducting electricity, and water boiling.What is the difference between a heterogeneous mixture and a homogeneous mixture? Give an example of each. Organo-main-group chemistry (B/Si/S) Asymmetric Sulfoxidation Sulfonium ylids Sulfoxonium Ylids Pummerer rearrangement Swern Oxidation Sulfoxide elimination Julia Olefination Hydroboration. 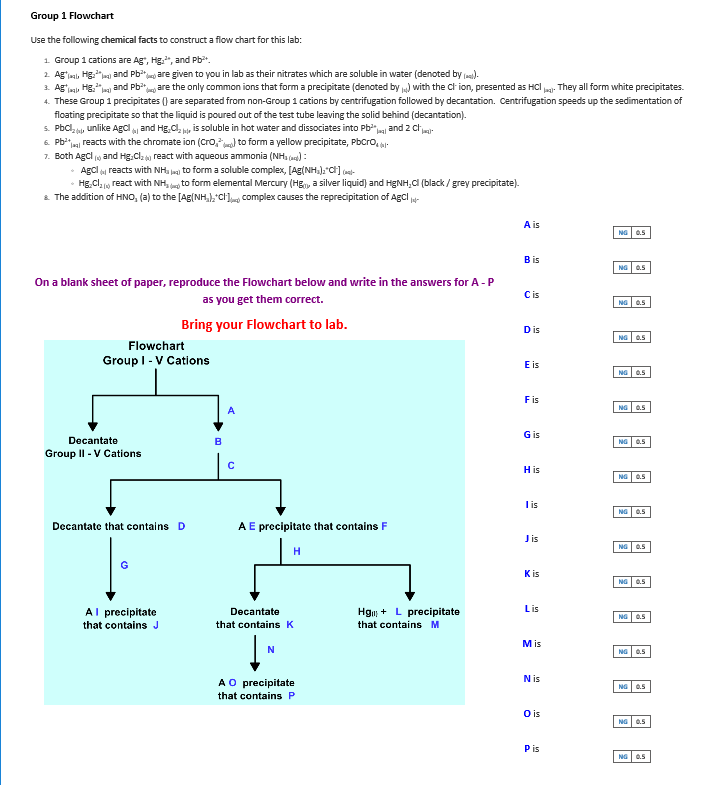
(iv) Their constituent particles (atoms, molecules or ions) have fixed positions and can only oscillate about their mean positions. (i) They have definite mass, volume and shape. Classify each as being element, compound, heterogeneous mixture, or homogeneous mixture (solution): air at Furman University, Furman Lake water, copper wire, and Furman tap water. 1.1 Characteristic Properties of the Solid State. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
